Heisenberg principle formula9/2/2023   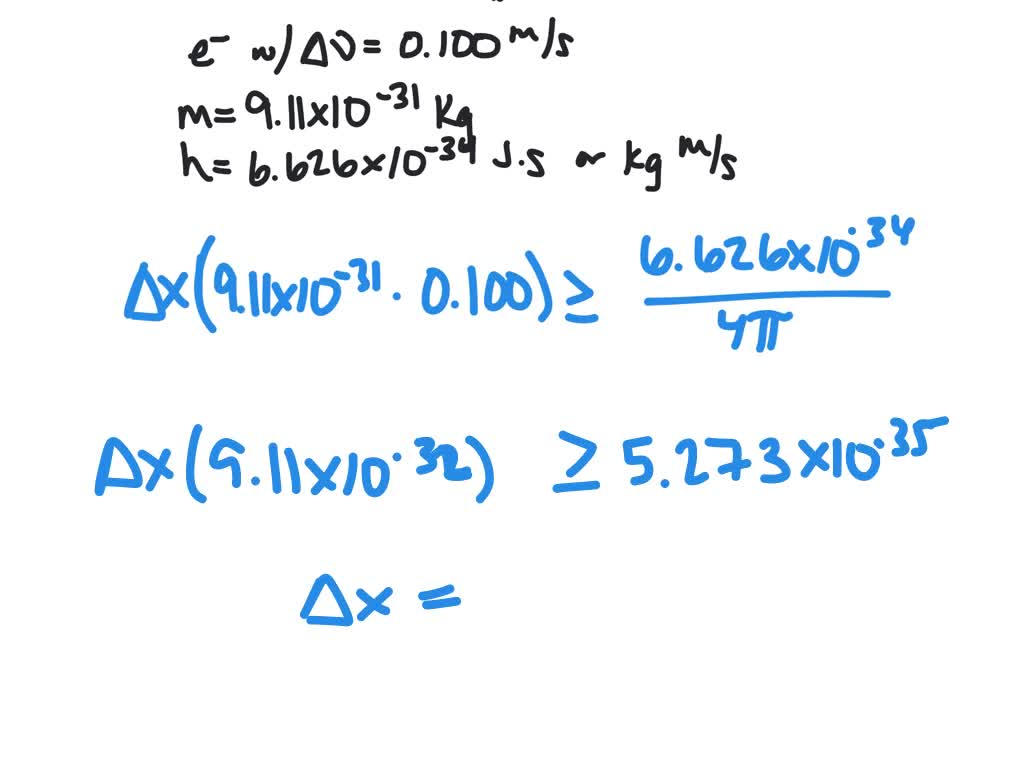
When position or momentum are accurately measured, it immediately suggests a higher inaccuracy in the measurement of the other quantity. Or, if you performe a large number of measurements, you'll obtain the uncertainty, $\Delta x$, is L/2 for the mean value x=0. The Uncertainty Principle gives a mathematically provable lower bound of the product of the uncertainty (error or deviation from a precise center of the. Heisenberg’s uncertainty principle formula can also be expressed as: xmv h/4. The wave packet is given by x-dependent wave function expressed in Equation A.2. If we instead carry out an experiment and successive measurements of two conjugate quantities, "each time returning the system to the $\Psi$ preceding the measurements", A and B, we obtain different values characterized by uncertainties $\Delta A$ and $\Delta B$ whose product will have an upper limit.Īs De Broglie said, we are therefore dealing with pre-measurement (in the first case) and post-measurement (in the second) uncertainty relations.įor instance, the infinite square well centered in the origin, the particle can occupy all positions between -L/2 and +L/2: so the average value is x=0 and the dispersion is L/2. Thus, g(k) is the Fourier transform of (r,0). For example, the values of the energy of a bound system are always discrete, and angular momentum components have values that take the form m, where m is either an integer or a half-integer, positive or negative. Heisenberg which states that higher the lifetime of a quantum. The observables discussed so far have had discrete sets of experimental values. Since you have a range of possibilities, the way naturally opens up for a statistical analysis: you have a distribution of values characterized by an average value and a dispersion, $\sigma$, around it.The product of the two $\sigma$ associated with the distribuitions of two coniugate osservables can't go below the value indicated in the HUP. Besides the equation (37), the is also an energy-time uncertainty relation given by W. before carrying out a measurement, an experiment) from a theoretical study of a physical system is, in general, a range of possible values. The statistical interpretation of quantum mechanics tells us that the "best" that we can know a priori (i.e. Keywords: Heisenbergs uncertainty principle, de Broglie interval wave, wave-particle duality, space-time, energy-momentum, Einsteins field equations. Development of full quantum mechanical theory edit Werner Heisenberg used the idea that since classical physics is correct when it applies to phenomena in the world of things larger than atoms and molecules, it must stand as a special case of a more inclusive quantum theoretical model. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
